Categories
Chembio Diagnostic # 60-9507-0 / 970-NI
RAPID TEST, SURE-CHECK HIV 1/2ASSAY 25 FINGER STICK TESTS PER KIT
$ 156.00 $ 156.00
Log in or create an account to proceed with checkout.
How Can We Help? Fast answers on bulk discounts, same-day shipping, and other questions for our team
Request Product Info
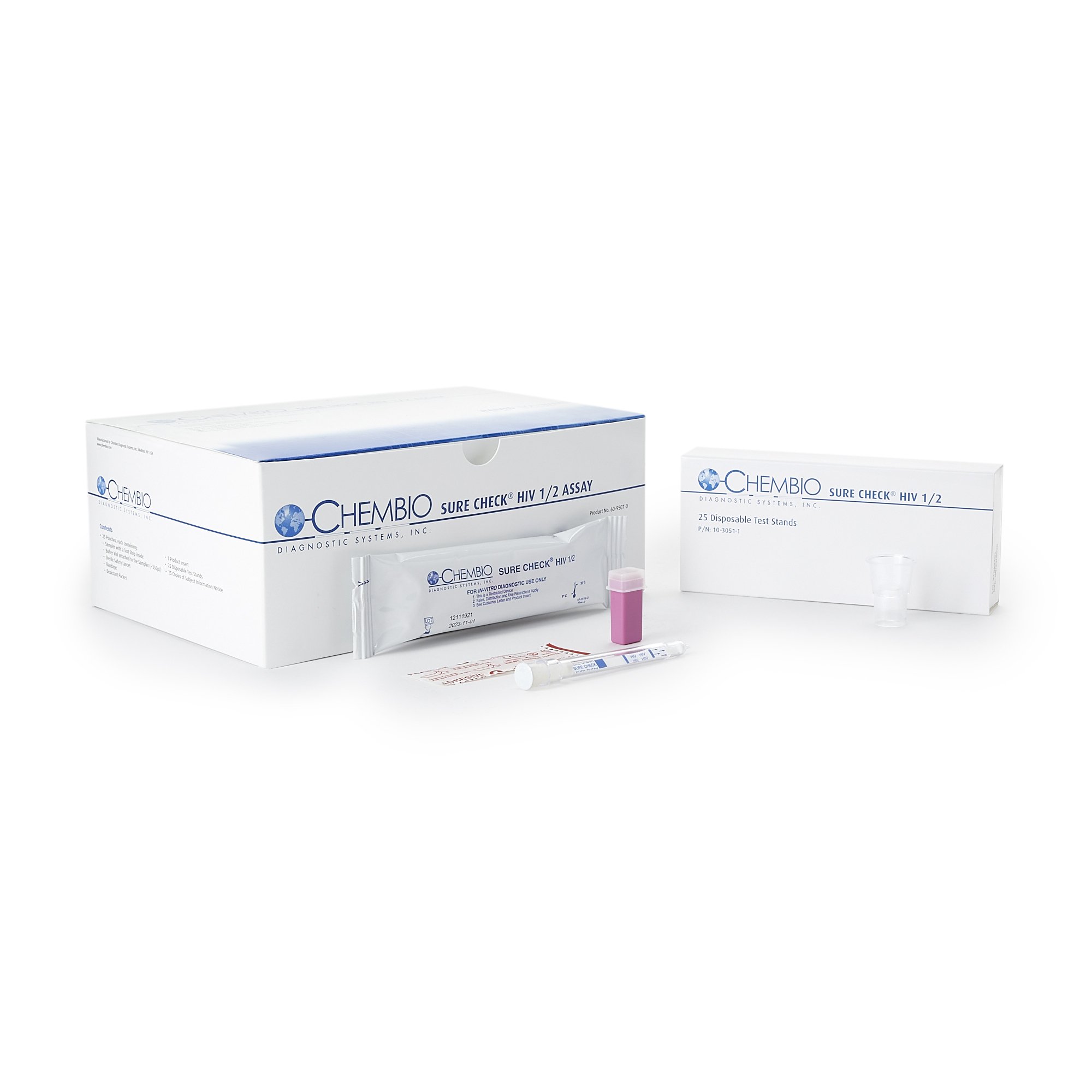
Chembio Diagnostic 60-9507-0 - Sure-Check HIV 1/2 Assay Rapid Test Kit, CLIA Waived, 25 Fingerstick Tests
The Chembio Diagnostic 60-9507-0 Sure-Check HIV 1/2 Assay is a critical tool for rapid, qualitative detection of antibodies to Human Immunodeficiency Virus Type 1 (HIV-1) and Type 2 (HIV-2). Designed for professional healthcare environments, this CLIA Waived rapid test kit provides efficient point-of-care screening, delivering reliable results in minutes.
Optimized for institutional workflows, each kit contains 25 individually packaged tests, facilitating standardized clinical use and streamlined inventory management within diverse healthcare settings.
Product Specifications
- SKU: 60-9507-0
- Brand: Chembio Diagnostic
- Model / Reference: Sure-Check® HIV 1/2 Assay
- Packaging: 25 Tests per Kit
- Application: Rapid detection of HIV-1/2 antibodies
- Certifications / Standards: CLIA Waived (for whole blood fingerstick samples); Buy American Act (BAA) Compliant; Trade Agreement Act (TAA) Compliant
- HCPCS / UNSPSC: 41116144
- Availability: In Stock
Key Features
- Integrated Design – Self-contained, single-use collection and testing device simplifies the rapid detection of HIV-1 and HIV-2 antibodies.
- Multiple Sample Compatibility – Suitable for use with whole blood (fingerstick or venipuncture), serum, and plasma samples, offering clinical flexibility.
- CLIA Waived for Whole Blood – Enables point-of-care testing in CLIA-waived settings when using fingerstick whole blood samples.
- Fast, Visual Results – Delivers clear, visually interpretable results in just 15 minutes, supporting timely clinical decisions.
- Comprehensive Fingerstick Kit – Each kit contains 25 individually pouched test devices, disposable test stands, buffer vials, sterile safety lancets, bandages, subject information notices, and quick reference guides for complete fingerstick testing.
- Compliant Procurement – Qualifies as Buy American Act (BAA) and Trade Agreement Act (TAA) compliant, simplifying institutional purchasing.
Suggested Applications
- Routine HIV screening in primary care settings
- Emergency department evaluations for rapid assessment
- Antenatal screening programs in obstetrics
- Sexual health clinics for point-of-care diagnostics
- Mobile health units requiring quick, reliable results
Industries of Use
Hospitals, Clinical Laboratories, Urgent Care Centers, Sexual Health Clinics, Physician Offices, Public Health Programs, and Long-Term Care Facilities.
The Chembio Diagnostic Sure-Check HIV 1/2 Assay offers healthcare organizations a dependable, compliant, and efficient solution for HIV-1/2 antibody screening. Its rapid results and CLIA Waived status for fingerstick samples make it ideal for high-volume clinical settings, ensuring seamless integration into existing supply chains and contributing to comprehensive patient care.