Categories
Chembio Diagnostic # 60-9549-0 / 1329-NI
RAPID TEST HIV CNTRL 0-2
$ 150.33 $ 150.33
Log in or create an account to proceed with checkout.
How Can We Help? Fast answers on bulk discounts, same-day shipping, and other questions for our team
Request Product Info
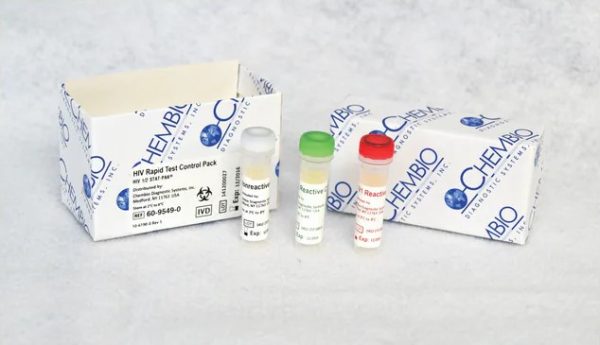
Chembio Diagnostic 60-9549-0 - STAT-PAK HIV-1/2 Rapid Test Control Set, Nonreactive/Reactive, 3 x 0.25 mL Vials
This Chembio Diagnostic STAT-PAK HIV-1/2 Rapid Test Control Set (60-9549-0) is engineered for stringent quality assurance in professional healthcare environments. It provides essential nonreactive and reactive controls for verifying the accuracy and reliability of HIV-1/2 rapid diagnostic assays, ensuring consistent and dependable test results for institutional buyers.
Product Specifications
- SKU: 60-9549-0
- Brand: Chembio Diagnostic
- Model / Reference: STAT-PAK HIV-1/2
- Size / Dimensions: Volume: 3 x 0.25 mL
- Packaging: 3 Vials (0.25 mL each), containing HIV-1 Reactive, HIV-2 Reactive, and Nonreactive controls; packaged with product insert
- Application: HIV-1/2 Rapid Test Control Set
- Material: Defibrinated Human Plasma
- HCPCS / UNSPSC: HCPCS: G0435 / UNSPSC: 41116145
- Availability: Ships within 1-2 business days
Key Features
- Comprehensive Control Set – Includes HIV-1 Reactive, HIV-2 Reactive, and Nonreactive controls for complete assay verification.
- Optimized for HIV-1/2 Rapid Tests – Specifically designed for use with Chembio Diagnostic Sure Check HIV 1/2 and HIV 1/2 Stat-Pak Assays.
- Liquid Form Controls – Supplied in convenient 0.25 mL vials for easy handling and precise application in laboratory settings.
- Essential Quality Assurance – Facilitates robust quality control protocols, critical for maintaining diagnostic accuracy and clinical reliability.
- Refrigeration Required – Ensures product integrity and stability through proper cold storage, adhering to clinical best practices.
Suggested Applications
- Routine quality control for HIV-1/2 rapid diagnostic testing in clinical laboratories.
- Verification of new lots or shipments of HIV-1/2 rapid test kits in hospitals and clinics.
- Training and competency assessment for laboratory personnel performing HIV-1/2 rapid assays.
- Ensuring compliance with regulatory and accreditation standards for diagnostic testing.
- Post-maintenance or troubleshooting verification of rapid test devices.
Industries of Use
Hospitals, Clinical Laboratories, Urgent Care Centers, Surgery Centers, Public Health Clinics, Long-Term Care Facilities.
The Chembio Diagnostic STAT-PAK HIV-1/2 Rapid Test Control Set is an indispensable component for robust quality control within professional healthcare supply chains. This control product supports standardized diagnostic workflows and helps procurement managers ensure the sustained accuracy and reliability of HIV-1/2 rapid testing across their institutional networks.